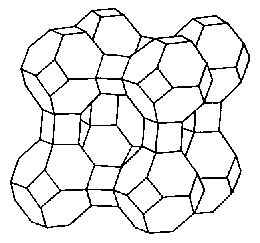
Type A Zeolite
The Molecular Sieves used for Carbon Dioxide Control are Zeolites, crystalline materials composed of silicon and aluminum. The crystal structure is based upon repeating units consisting of a silicon atom (+4 valence) surrounded by four oxygen atoms (-2 valence) in a tetrahedral configuration. Each oxygen is shared by two Si atoms, giving the tetrahedra net valences of zero. When aluminum (with a valence of +3) is substituted in the tetrahedra a net charge defect of -1 occurs. This gives rise to the cation exchange properties of zeolites. Another interesting property of zeolites is their very uniform pore sizes which occur as a consequence of their unique crystal structures. Zeolites have both very high porosities and very well defined pore sizes, hence their use as Molecular Sieves. Both crystal and pore geometries are illustrated below for Type A and Type X zeolites.

Type A Zeolite
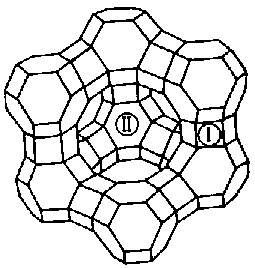
Type X Zeolite
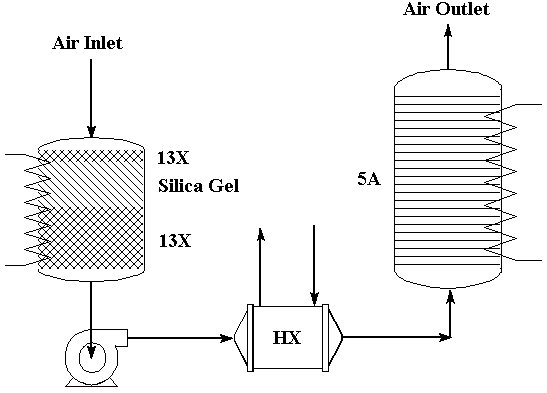
The Air Revitalization System for ISSA includes a regnerative Four Bed Molecular Sieve system for the removal of CO2 from cabin air. The system uses a combination of silica gel and Molecular Sieve 13X to remove water vapor, followed by Molecular Sieve 5A to sorb CO2. This combination is necessary because water vapor seriously inhibits the adsorption of carbon dioxide by 5A. The system is thermally regenerable. While two beds are in the sorption mode, the other two beds are undergoing regeneration. A system of this kind is illustrated schematically below.
Author: Tugrul Sezen BACK
TO SPACE SETTLEMENT HOME PAGE
[email protected]
|
Curator: Al Globus If you find any errors on this page contact Al Globus. |
 |
This site was hosted by the NASA Ames Research Center from 1994-2018 and is now hosted by:
