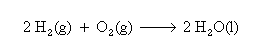
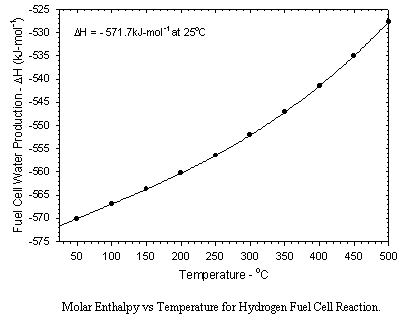
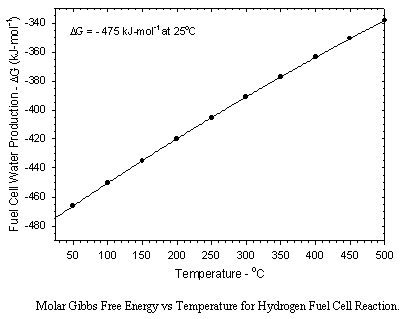
Aboard the Shuttle Orbiter water is generated using a hydrogen-oxygen fuel cell which produces both electricity and very pure water. Water can also be produced by reduction of CO2 by hydrogen over a suitable catalyst. The Sabatier and Bosch processes are two Carbon Dioxide Reduction Systems which have been studied extensively. The Sabatier method has been selected for International Space Station (ISS). These three systems are presented below:
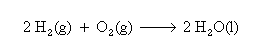


Current technology for the reduction of carbon dioxide to produce water aboard the International Space Station (ISS) is embodied in the Sabatier Reactor.
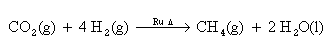
For each mole of carbon dioxide reduced, this system produces two moles of water and one mole of methane. The methane is vented to space. For this reason, only half of the hydrogen consumed in this reaction can be recovered by electrolysis. The Sabatier reaction utilizes an alumina supported ruthenium catalyst and, in comparison to the Bosch reaction, has the advantage of greater thermodynamic favorability and higher reaction rates.
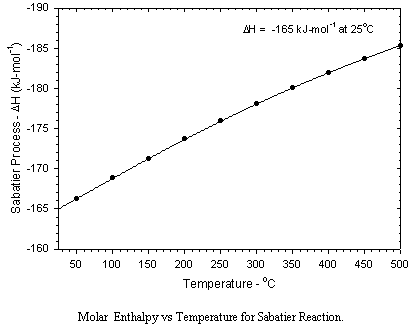
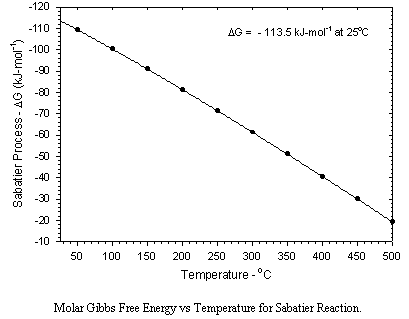
The primary disadvantage of the Sabatier process is that twice as much hydrogen is consumed as can be recovered by the electrolysis of the produced water. This results in a substantial perturbation in the material balance for the production of oxygen from carbon dioxide. Using the Sabatier process either the hydrogen imbalance must be compensated for by resupply, or not all of the carbon dioxide can be processed.
The Bosch reaction produces only water and elemental carbon, allowing all of the hydrogen consumed to be recovered by electrolysis. This makes closure of the oxygen - carbon dioxide - water - oxygen loop possible.
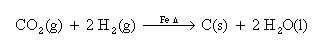
The net reaction produces heat (2.3x103 J/g of carbon produced at 650C), and is the result of two successive reactions, one fast,
and a second rate controlling slow reaction.
The primary problems associated with the Bosch reactor are slow reaction rates and the generation of particulate carbon which fouls the catalyst surface and which plugs the catalyst bed. As shown below, the thermodynamics are also somewhat less favorable than those for the Sabatier Reaction.
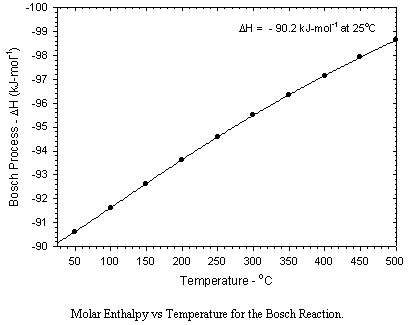
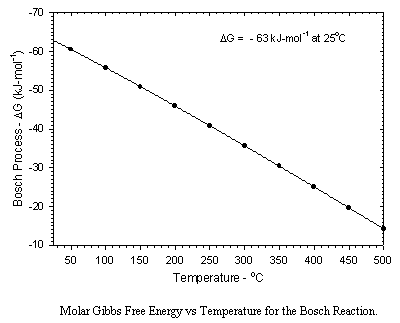
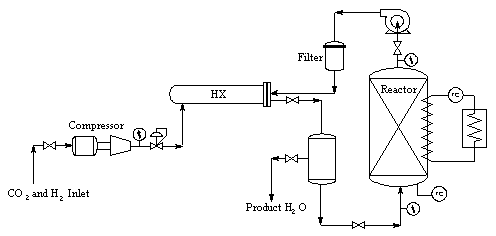
Catalysts are critical to the Bosch process, due to the intrinsically slow kinetics. Iron, cobalt, and nickel catalyze the Bosch reaction. Minor additions of ruthenium significantly improve kinetics. The most commonly used catalyst is "steel wool" due to its relatively high exposed active surface. Significant surface reactions include the formation of carbide intermediates as well as oxide layers which poison catalytic activity. Typical reaction temperatures are in the range of 450 to 600C. Even at these relatively high temperatures single pass reduction efficiencies are typically below 10%, hence practical reactor designs require recycle operation. A more significant disadvantage is the inactivation of catalyst surfaces and the plugging of the reactor caused by a buildup of particulate carbon. While greater efficiencies in terms of material balances aboard spacecraft are possible using the Bosch reaction, much further development work will be required before the system is competitive with the Sabatier process from a practical standpoint.
Author: Tugrul Sezen BACK
TO SPACE SETTLEMENT HOME PAGE
[email protected]
|
Curator: Al Globus If you find any errors on this page contact Al Globus. |
 |
This site was hosted by the NASA Ames Research Center from 1994-2018 and is now hosted by:
